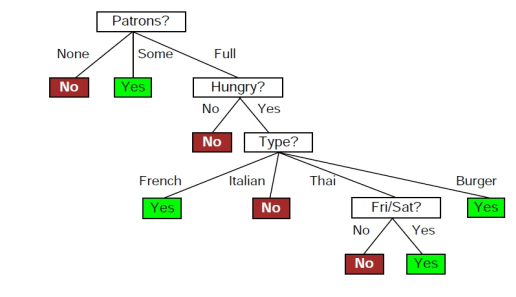
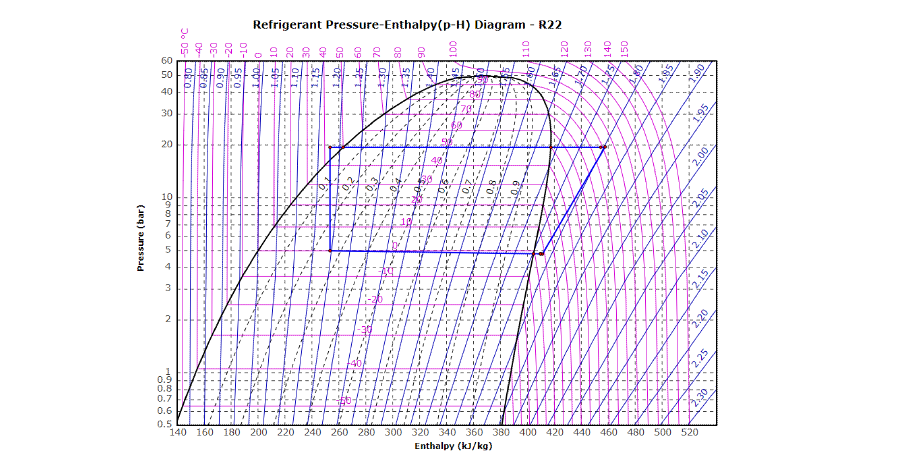
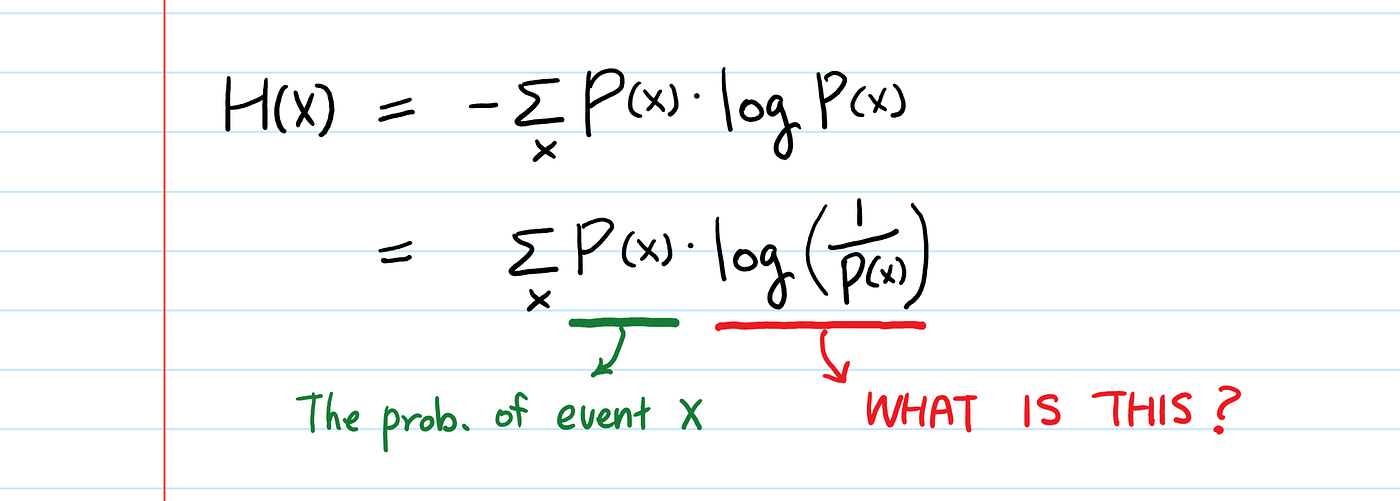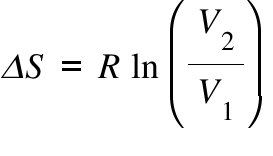Calculate the standard entropy change for a reaction X harr Y, If the value of DeltaH^(@) = 28.40 kJ and equilibrium constant is 1.8 xx 10^(-7) at 298 K.

Entropy Calculation, Information Gain & Decision Tree Learning | by Badiuzzaman Pranto | Analytics Vidhya | Medium